

Our Mission at GNT Animal Health
We aim to provide products that go beyond merely treating and controlling animal diseases.


R&D history
The background of GedaCure® :
The world’s first canine cognitive dysfunction syndrome treatment.
The world’s first canine cognitive dysfunction syndrome treatment.
Development of crisdesalazine, the active pharmaceutical ingredient of GedaCure®
GedaCure®, developed by GNT Pharma, is the world's first canine cognitive dysfunction syndrome treatment. About a decade
ago, GNT Pharma has developed a library of hundreds of compounds as candidates for the treatment of human Alzheimer's Disease (AD)
and studied their pharmacological actions and safety with the support of “the 21st century Brain Frontier R&D project” from MOST
(the Ministry of Science & Technology) and “the Large National R&D Commercialization Program” of MHWS (Ministry of Health and
Welfare) in South Korea. Among them, we found crisdesalazine, a new drug candidate with excellent safety and outstanding efficacy to
protect brain cell death by simultaneously removing inflammation and free radicals which are known as the main causes of AD.
-
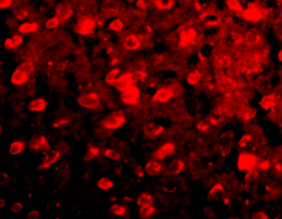
 APP/PS1
APP/PS1
-
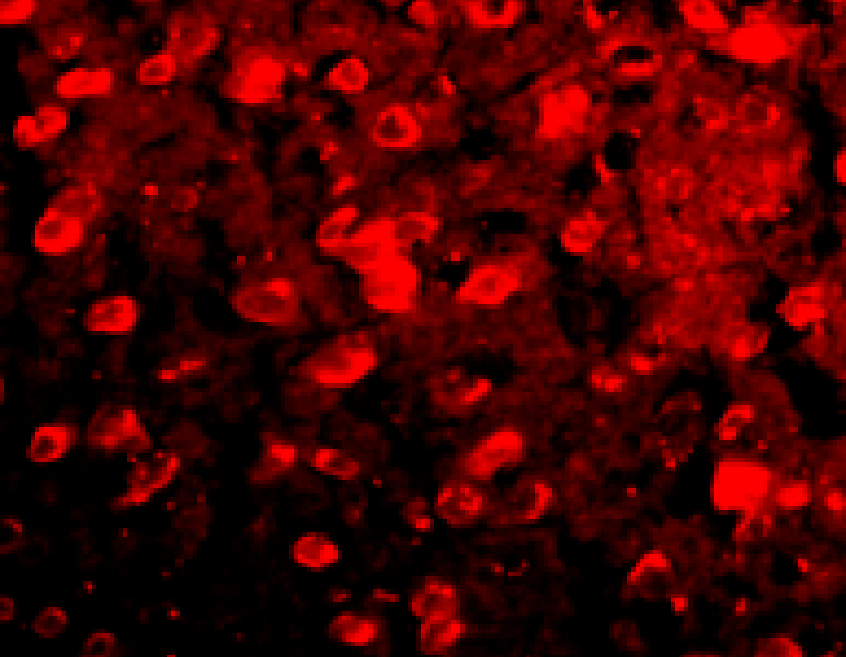
 + crisdesalazine
+ crisdesalazine
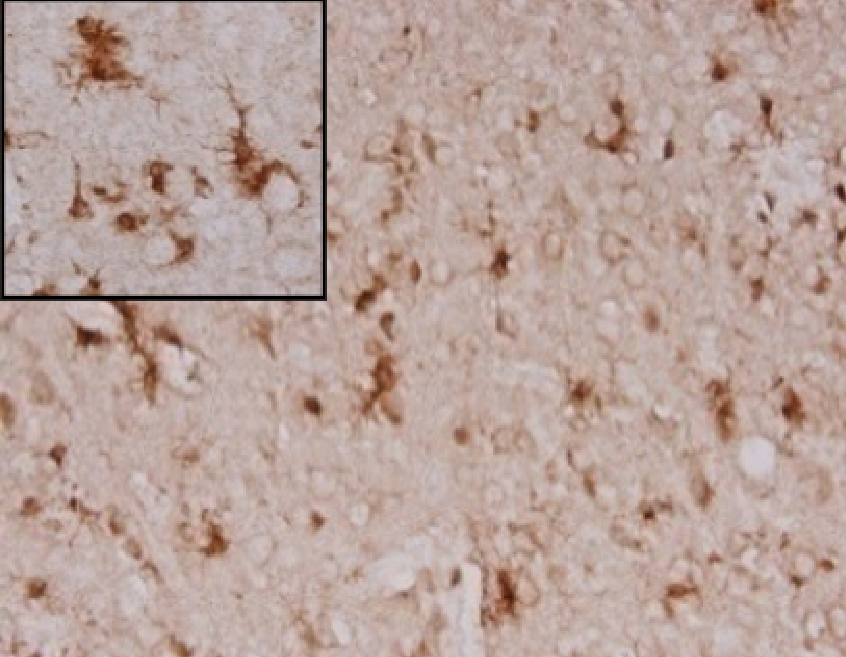
Anti-oxidative effect in AD animal model
-
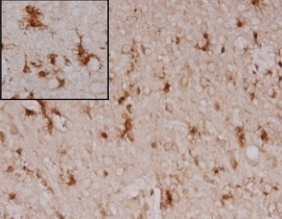
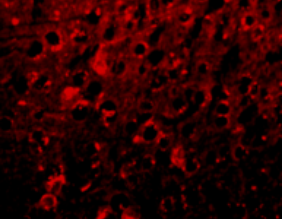
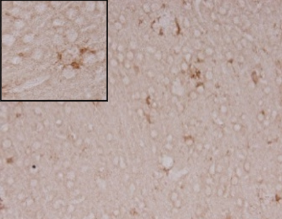
 APP/PS1
APP/PS1
-
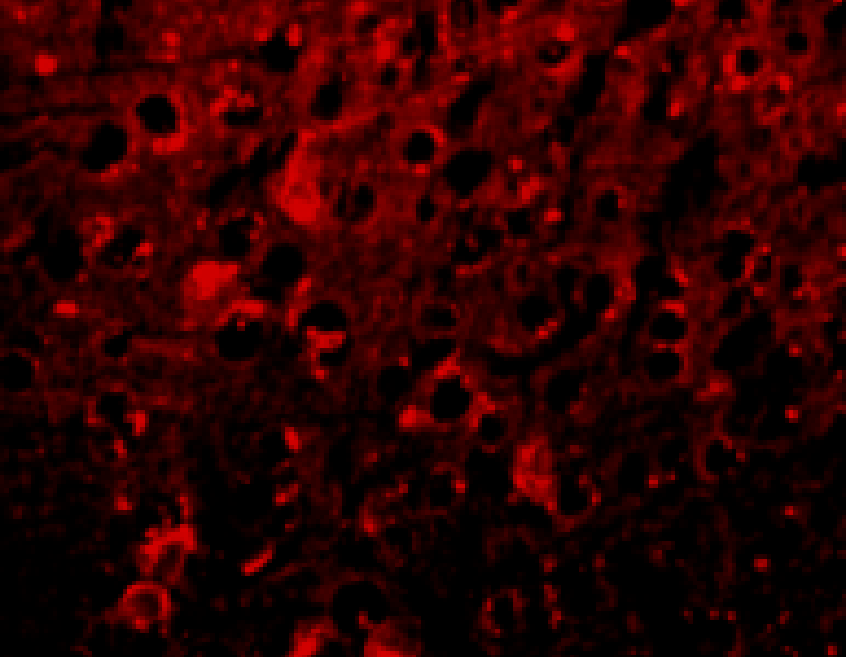
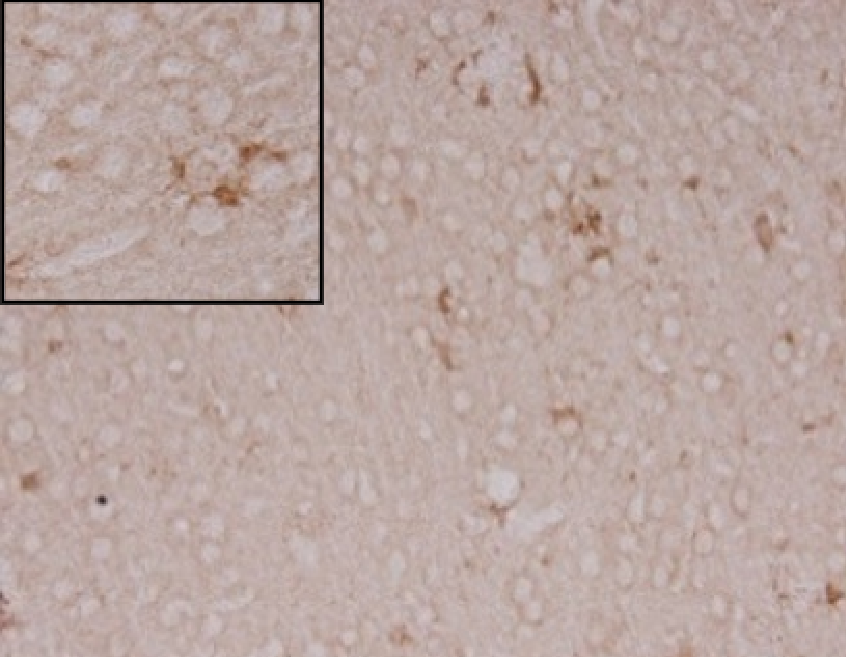 + crisdesalazine
+ crisdesalazine
Anti-inflammatory effect in AD animal model
The efficacy of crisdesalazine in mouse models of AD
To verify the efficacy of crisdesalazine on AD, we have tested using various genetically modified transgenic mouse models such as
“Tg2576 and APP/PS1 mouse” which are commonly used in AD studies and “Tg-betaCTF99/B6 mouse” that GNT Pharma developed. As a result,
long-term administration of crisdesalazine has proven to reduce amyloid plaque, a typical pathological symptom of AD, as well as to
suppress oxidative stress and inflammation, protect the blood-brain-barrier damage, prevent neuronal cell death, and improve
cognitive function. In a GiD mouse model with severely overactivated reactive astrocytes, which was recently proposed as a new AD
model, it was proven that crisdesalazine has the effects of preventing brain atrophy and improving cognitive function by inhibiting
the toxic production of hydrogen peroxide (H2O2) and anti-inflammatory effects. These results suggest that
crisdesalazine has sufficient potential as a treatment of AD.


Similarity between CCDS and AD
By the time we found the possibility of crisdesalazine as a treatment of AD, our researchers noticed that dogs also have a dementia
and its symptoms were almost similar to those of human AD. The official name of canine dementia is called Canine Cognitive
Dysfunction Syndrome (CCDS), which represents almost the same pathological characteristics as typical hallmarks of human AD such as
amyloid plaque accumulation, neurofibrillary tangle formation, neuronal cell death, and cognitive impairment.


Approval and launching GedaCure®
In February 2020, GNT Pharma submitted the application of
GedaCure® for the approval of veterinary drug based on the
results of the clinical trials. Finally, GedaCure® got approved
as the first new CCDS drug of South Korea in February
2021. GAH will actively target overseas markets such as the
U.S., Europe, Japan, and China in the future to expand its
global market share. We wish many dogs and owners to
lead a better life with GedaCure®.
Verifying efficacy and safety
of crisdesalazine in dogs with CCDS
of crisdesalazine in dogs with CCDS
Therefore, taking into consideration the similarities between AD and CCDS and the large unmet needs of the canine CCDS market, we
have established a GNT Animal Health (GAH) business unit and started to develop crisdesalazine as the world's first canine
cognitive dysfunction syndrome treatment. In 2018, we conducted a pilot study at Irion Animal Hospital and observed significant
efficacy of crisdesalazine. In 2019, we appointed Yoon Hwa-young, Professor of the Department of Veterinary Internal Medicine,
College of Veterinary Medicine at Seoul National University, as a principal investigator and successfully completed a phase III
clinical trial for marketing approval enrolling 48 dogs with CCDS through cooperation with five animal hospitals in South Korea.
Clinical sites
GAH has successfully developed GedaCure®
through clinical studies with
domestic animal hospitals and researchers.
domestic animal hospitals and researchers.

